Record extremely low temperatures well below freezing point with our TRIL-8 temperature data logger. This logger can be configured multiple times for different recording intervals and alarm thresholds between -80 °C and +40 °C.

The SRIL-8 temperature logger is designed for one-time configuration, making it ideal as a cost-effective logger for monitoring transport and storage conditions well below-freezing temperatures.
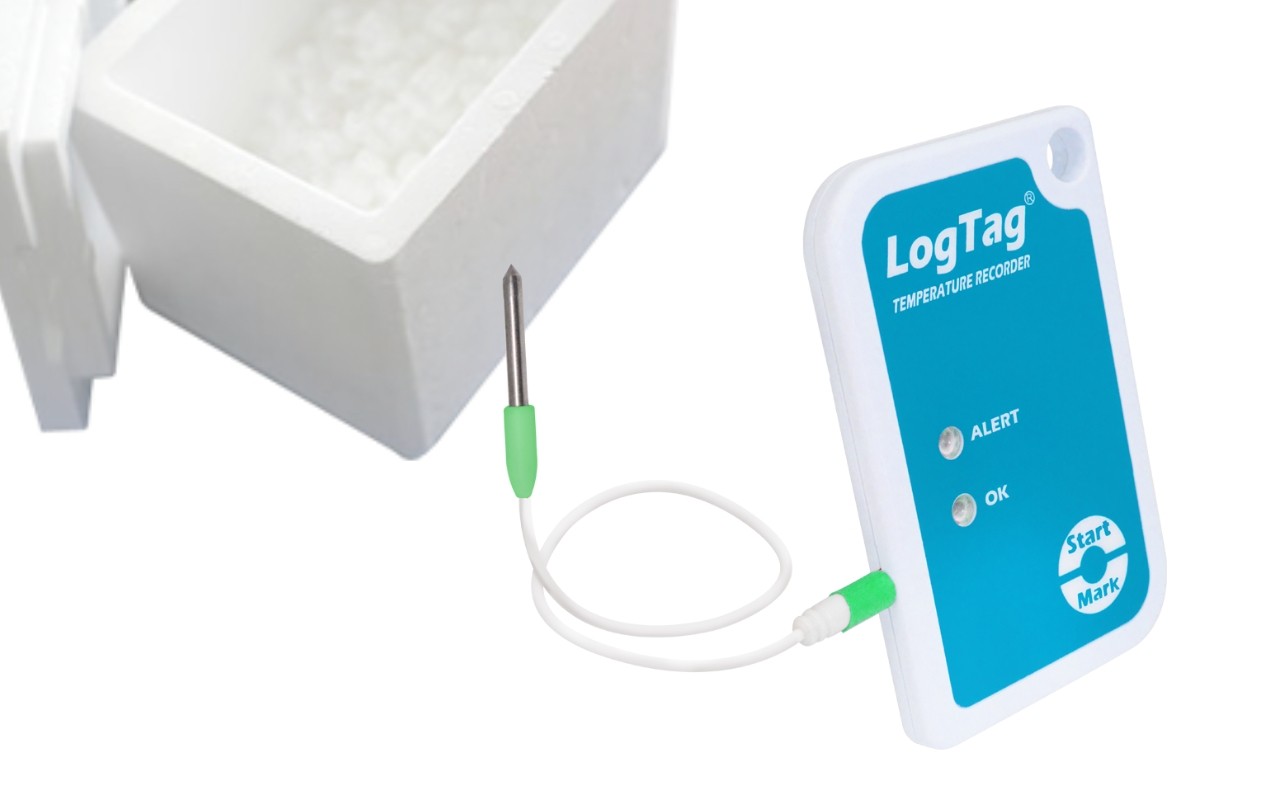
Dhe TREL-8 data logger has been specially designed for use at ultra-low temperatures and is characterized by its external, food-safe probe. This allows for example the measurement of temperature data in a chest freezer without the need to remove the probe or to open the chest. Ideal for temperature monitoring during vaccine storage.
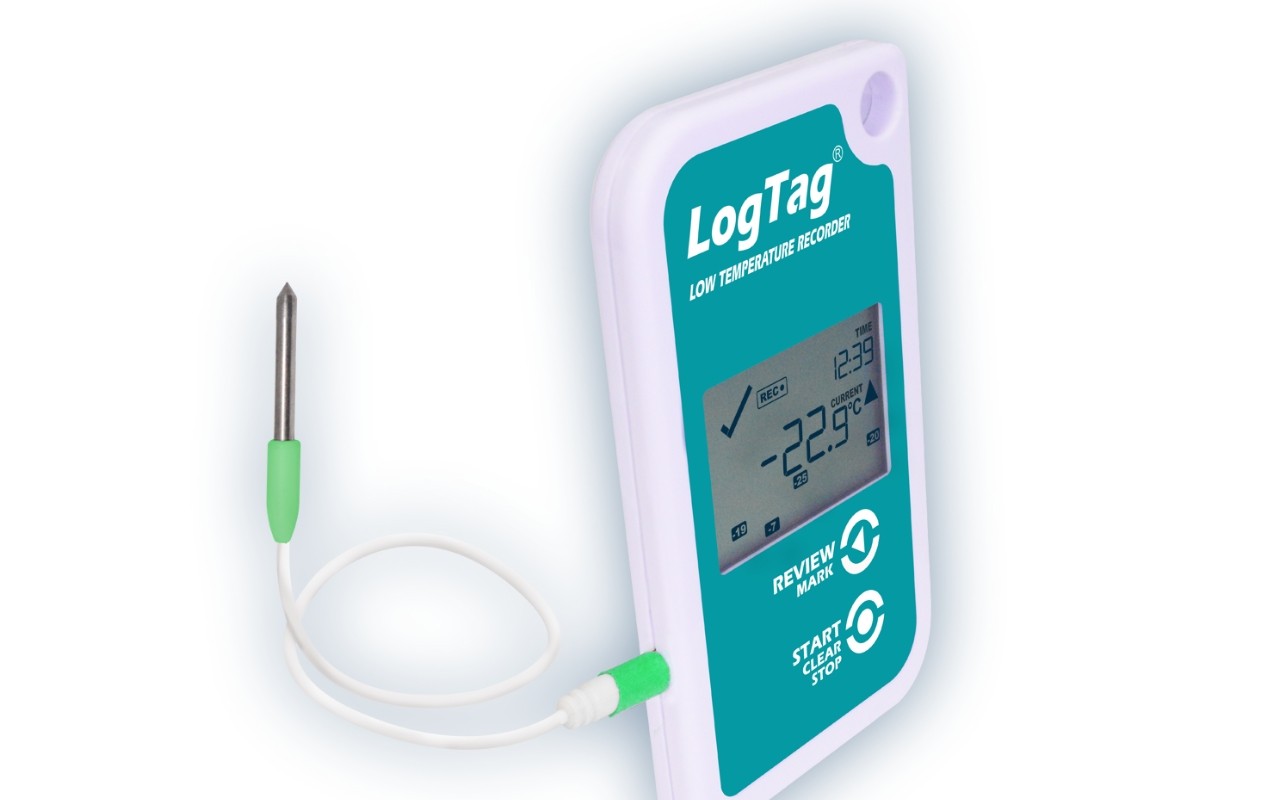
With the TREL30-16 data logger with 30-day statistics display and alarm, you have your ultra-low temperatures firmly under control. Designed specifically for use in ultra-low freezing temperatures, it is ideally suited for monitoring low-temperature storage facilities.

The LogTag® UTREL-16F temperature data logger can record up to 16,000 readings in a measuring range from -90 °C to +40 °C via the external sensor. The UTREL-16F data logger is a cost-effective and reliable LOW-temperature USB data logger with display

Temperature measurement where access requires a remote probe and data needs to be accessed without a PC. The LogTag UTREL30-16 DUAL provides the facility to create a detailed PDF report without need for extra accessories and proprietary software only with the supplied USB cable. The display is designed to show ‘at a glance’ if temperature excursions have occurred during the current day and up to the previous 30 days.

The LogTag® UTREL30-WiFi DUAL is a 30 day ultra low temperature wifi logger with display, The LogTag® UTREL30-WiFi DUAL temperature data logger automatically uploads real-time temperature data to your LogTag® Online cloud account via your existing wireless network.
The large, easy-to-read display, is designed to show ‘at a glance’ if temperature excursions have occurred during the current day and up to the previous 30 days.
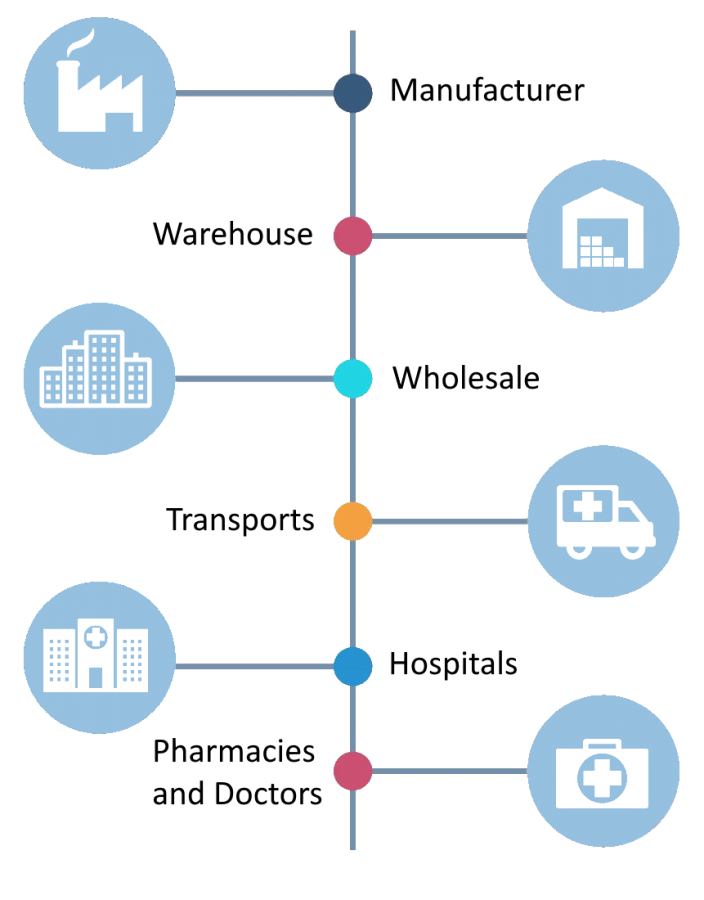
Vaccines are key components in disease prevention and control. It is of the utmost importance that vaccines are handled carefully, kept in the right conditions and at the right temperatures at all times. The quality of a vaccine is everyone's joint responsibility, from the manufacture to the administration of the vaccine.