
The LogTag Digital Signatures suite has been specifically developed to support the FDA title 21 CFR part 11 which often is obligatory in the pharmaceutical, biotech and medical device industry.
In these software add-ons, authenticated users can digitally sign a set of recordings with a given set of digital signatures that has been allocated to those users.
A Digital Signature is registered with the recordings and contains information associated with the signing that clearly indicates all of the following:
- The printed name of the signer
- The date and time when the signature was executed
- The meaning (such as review, approval, responsibility, or authorship) associated with the signature
Digital signatures remain permanently stored with the Logtag data file.
Authenticated Users are identified by unique usernames and passwords.
LogTag uses a “client-server” approach for authenticating users and digital signatures.
LogTag uses three pieces of software for authenticating users and digital signatures: the standard software «LogTag Analyzer» plus two add-on software called «LogTag User Server» and «LogTag Event Viewer»
Diagram of system
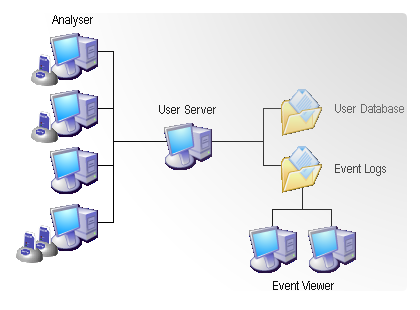
LogTag Analyzer
This is the standard software for reading and configuring LogTag loggers and runs on computers that are reading, displaying and storing logger data.
LogTag User Server
User Server is normally run on a server in a networked computer system but can be run on the same computer as Analyzer provided security issues are observed.
The purpose of the User Server software is to provide access to the user & signature database to all appropriately configured Analyzer clients and to maintain the audit event log of user activities.
LogTag Event Viewer
Allows viewing of the audit events. It can be run on the same computer as LogTag User Server and/or other workstations provided those workstations have network access to the folders on the user server computer in which the event audit log files are stored.
Please refer to the LogTag Digital Signatures User Guide for further detail.
Click here to download latest release versions of LogTag User Server & LogTag Event viewer
